Nearly 1 covid vaccine in every 7-8 people worldwide was administered using HMD’s syringeHMD had supplied 921.7 million syringes to the Government of India to facilitate over 2.2 billionVaccination in India till date.Hindustan Syringes & Medical Devices Ltd (HMD), one of the leading manufacturers
Georgia Embassy’s flagship education fair organized in Mumbai eyed inflated demand from Indian medical studentsThe second edition of the Georgia Education Fair organized in Mumbai focused on providing holistic information on medical and technology courses including MBBS, Dentistry, Data Science, Artificial Intelligence, and many among
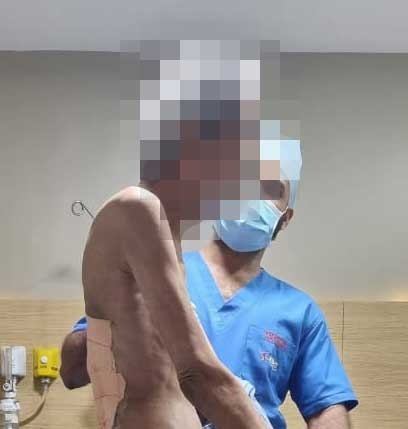
Ankylosing Spondylitis caused the problem* Complex surgery was done at KIMS Kondapur* Patient completely recovered after surgery An 80-year-old patient was suffering from a stiff neck and spine for the last 5 years. He is well-known in the society. But, he was unable to walk erect
The partnership will enable residents of Columbia Pacific Communities to avail Portea’s diverse home healthcare services at discounted rates. Portea will be the official Home Healthcare Partner of Columbia Pacific Communities for all its communities in India. Columbia Pacific Communities (CPC), India's largest and most
QOQ Q4 FY 23 vs. Q4 FY 22: The Financials have been prepared in accordance with Ind AS (‘Indian Accounting Standards’):The Revenue from Operations is Rs. 3.40 Cr. in Q4 FY 23 as against Rs. 2.61 Cr. in Q4 FY 22,which is an increase of
AstraZeneca Pharma India Ltd. (AstraZeneca India), a leading biopharmaceutical company, announced the appointment of Bhavana Agrawal as Chief Finance Officer (CFO) with effect from October 1, 2023, following a transition period. She succeeds Rajesh Marwaha, who is retiring as CFO and full-time director of the
Organ donation is a noble act that can save multiple lives. It is a gift that can be given by anyone, regardless of age, race, or medical history. However, there is a shortage of organs available for transplant, and many people die waiting for a
A team headed by Dr Deepak Gautam, Director of Orthopedic disciplines at Medicover Hospitals, Navi Mumbai successfully managed a 56-year-old patient by performing a total hip replacement using the latest minimally invasive procedure with a direct anterior hip approach for the first time in Navi
82.8% women struggle to focus on work due to menstrual periods or pain associated with it India’s leading feminine hygiene brand, Everteen, has released findings of its 8th Annual Everteen Menstrual Hygiene Survey 2023. Everteen is one of the partners for #MHDay2023, which is also supported
Concept Medical, a globally recognised leader in innovative medical devices, has achieved a remarkable milestone by becoming the first medical company to receive its fourth Investigational Device Exemption (IDE) approval from the United States Food and Drug Administration (USFDA) to treat Superficial Femoral Arteries (SFA).The