Freudenberg Expands Bioprocessing Capabilities at CPHI & PMEC India
Bangalore, India – 25 November 2025 – Freudenberg Medical, a global manufacturer of Tubings and custom assemblies for Biopharmaceutical Industry and Medical devices, will showcase its products and services at India’s largest pharma expo -

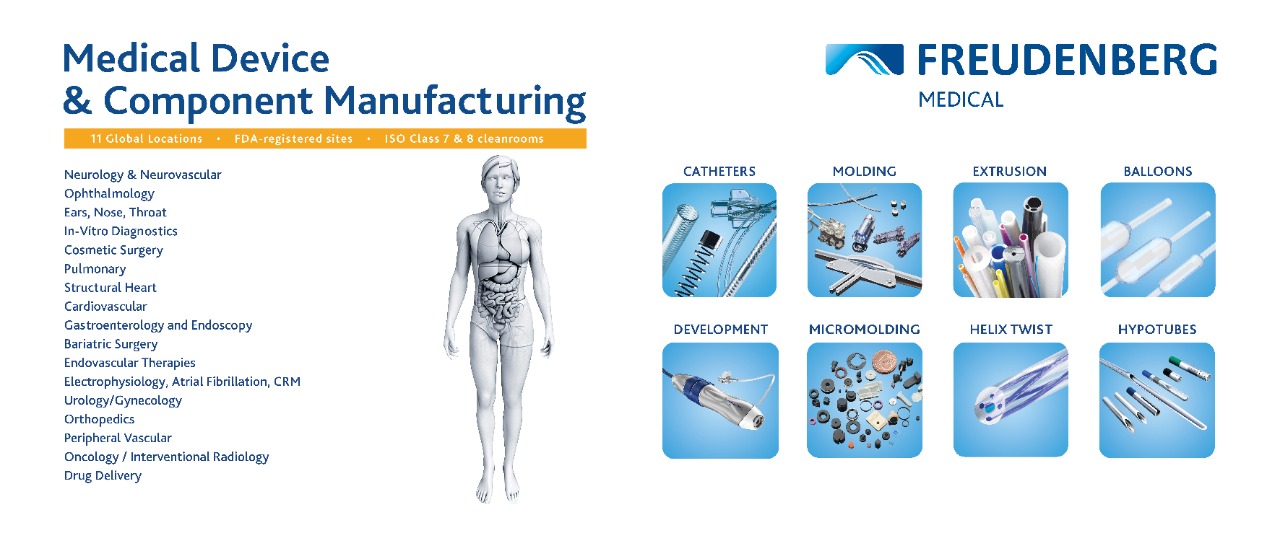
Bangalore, India – 25 November 2025 – Freudenberg Medical, a global manufacturer of Tubings and custom assemblies for Biopharmaceutical Industry and Medical devices, will showcase its products and services at India’s largest pharma expo – CPHI & PMEC India, held from 25 – 27 November 2025 at Booth 15.A22. Freudenberg manufactures high purity fluid handling products from two dedicated sites in the United States and Germany.
What’s new:
- Freudenberg Medical’s Biopharma Solutions Group has installed an industry first “CleanAssure” system: A new sanitary washing and drying workstation within the cleanroom that utilizes ultra-purified water for washing at a specified temperature range and then drying with filtered air. This system is designed and deployed for customers who need Single Use Assemblies and Tubings with added assurance for safety.
- Freudenberg has also expanded their capabilities for single-use assemblies with additional over molded silicone connections for bioprocessing and pharmaceutical manufacturing, these include T, Y and cross connectors. Over molded silicone connections offer an unobstructed smooth flow for a critical fluids path, preventing any potential chances of leakage and fluid entrapment into intra-spaces that may result while using standard fluid couplings, fittings, and connectors.
- Depending on customer’s requirements, Freudenberg also offers custom cut-length tubings, standard tubings & Single-Use Assemblies with Gamma sterilization.
Expansion
Freudenberg Medical opened its second production location in Costa Rica in September 2025. With US $25M investment, the new manufacturing operation is 50,000 square feet with three ISO Class 7 cleanrooms operational from day one. Plans have been announced to double the site to 100,000 square feet within the next three years — quadrupling Freudenberg Medical’s total footprint in Costa Rica.
Sustainability
Freudenberg Medical completed the successful global roll out of a state-of-the-art environmental management system and announced ISO 14001 certification for 11 global
locations. The achievement caps a global, multi-year effort to reduce environmental impact and a step towards reaching carbon neutrality of its manufacturing activities. In addition, two manufacturing operations have been using 100% green energy with all electricity purchased from renewable energy i.e. solar energy.
“Freudenberg Medical is a leader in sustainable manufacturing for the pharmaceutical and medical device industry,” said Falgun Jani, Business Head, Freudenberg Medical India. “Sustainability is an important guiding principle of the Freudenberg Group, and we are committed to protecting the environment and the communities in which we operate.”
Secure Supply Chain
Freudenberg’s identical manufacturing facilities – in the USA and Germany – ensure high quality cleanroom production of tubing, and a secure supply chain for customers. This reflects Freudenberg’s commitment to providing high purity and safe fluid transfer solutions. Freudenberg tubing is available off-the-shelf or with short lead time supported by a dedicated sales and service team in India & Germany that assures timely order processing and delivery.
Visit our website to explore our biopharma product portfolio and learn more.
Visit Freudenberg Medical & Freudenberg Filtration Technologies at CPhI & PMEC Expo in Delhi NCR, India, 25th -27th Nov at Stand 15.A22.