Minister Anupriya Patel Inaugurates National VRDL Conclave 2025; Launches IVD Validation Portal
New Delhi, September 11, 2025: Union Minister of State for Health and Family Welfare Smt. Anupriya Patel inaugurated the National Virus Research & Diagnostic Laboratory (VRDL) Conclave – 2025 in New Delhi today, marking a

New Delhi, September 11, 2025: Union Minister of State for Health and Family Welfare Smt. Anupriya Patel inaugurated the National Virus Research & Diagnostic Laboratory (VRDL) Conclave – 2025 in New Delhi today, marking a major milestone in India’s health research and diagnostics ecosystem.
The event was attended by Dr. Rajiv Bahl, Secretary, Department of Health Research (DHR) & DG, ICMR, and Dr. Rajeev Singh Raghuvanshi, Drug Controller General of India, along with senior health officials, scientists, and representatives from 165 VRDLs across the country.
VRDLs: “Sentinels of India’s Health Security”
Addressing participants, Smt. Patel praised the VRDL network for its role in safeguarding the nation during public health crises.
“Our VRDLs have stood as sentinels in protecting the country during the COVID-19 pandemic and beyond. Each laboratory, each innovation, each collaboration contributes to the vision of Viksit Bharat,” she said.
She highlighted that 16 VRDLs are now equipped with Bio-Safety Level-3 (BSL-3) facilities to study high-risk pathogens, while the National Institute of Virology (NIV), Pune continues as India’s only BSL-4 laboratory. Under the Ayushman Bharat Health Infrastructure Mission, four new regional NIVs are being established in Jabalpur, Dibrugarh, Bengaluru, and Jammu.
Launch of IVD Validation Portal
A major highlight of the conclave was the launch of the In-Vitro Diagnostics (IVD) validation portal and standardized protocols, developed jointly by ICMR and CDSCO. The platform will enable faster, transparent, and industry-friendly validation processes, strengthening India’s position in the global diagnostics market.
Innovations in TB and Tribal Health
Smt. Patel outlined India’s progress in tuberculosis elimination through innovations validated by ICMR:
Portable handheld X-ray machines deployed in remote villages under the 100-day TB campaign.
DeepCXR, an AI-based screening tool trained on over 75,000 X-rays, now freely available for mass TB screening.
CyTb skin test priced at just ₹199 for detecting latent TB.
PathoDetectTM for rapid and accurate molecular TB diagnosis.
Modified BPaL regimen showing up to 90% cure rates in drug-resistant TB cases.
In tribal health, she noted that the National Sickle Cell Anaemia Elimination Mission has brought down the cost of diagnostic tests from ₹300 to ₹28, improving accessibility and affordability.
Evidence-Based Policymaking
The Minister also emphasized the role of Health Technology Assessment in India (HTAIn) in revising 855 health benefit packages under Ayushman Bharat–PMJAY, based on data from 86 hospitals. This has optimized both cost and quality of healthcare delivery.
Release of VRDL Bulletin
Smt. Patel released the first VRDL Bulletin, a quarterly publication to track viral pathogens, seasonal trends, and outbreaks, enabling timely policy interventions and efficient resource allocation.
Awards to Top-Performing VRDLs
A total of 25 VRDLs were felicitated for excellence in diagnostics—8 under the Gold category and 17 under the Silver category. These included premier institutes such as AIIMS Bhopal, JIPMER Puducherry, Government Medical College Amritsar, and AFMC Pune.
One Health Mission
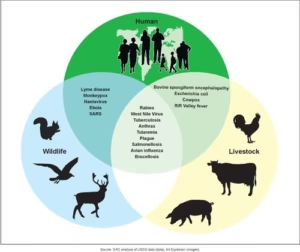
Underscoring India’s holistic approach, the Minister highlighted the National One Health Mission, which brings together 13 departments under a single framework for the first time globally. Ten VRDLs have been integrated into a National BSL-3 Network as part of this initiative.
“From early detection of outbreaks to indigenous test validations, from TB and sickle cell innovations to the One Health Mission, India is building a resilient, self-reliant, and globally relevant health research ecosystem,” she noted.
Leaders’ Perspectives
Dr. Rajiv Bahl, DG-ICMR: “The VRDL network will act as India’s impenetrable bio-defence, enabling early detection and rapid response to emerging infections.”
Dr. Rajeev Singh Raghuvanshi, DCGI: “The 39 protocols for high-risk IVDs are the first global example of collaboration between a national regulator and a national research body.”
The conclave concluded with recognition of the VRDLs’ contributions to strengthening India’s diagnostic and surveillance capabilities, reaffirming the country’s commitment to building a robust, future-ready public health infrastructure.