India’s ambitious efforts to lower its reliance on medical device imports are being curbed by stringent Production Linked Incentive (PLI) scheme requirements and persistent infrastructure underutilization, according to a recent report by the Boston Consulting Group (BCG). The report, titled “Unlocking ‘India for the World’
Union Minister for Road Transport & Highways, Shri Nitin Gadkari dedicated to the people of Nagpur a state-of-the-art charitable diagnostic centre envisioned and established under his leadership to serve the city’s underprivileged communities. The Swargiya Bhanutai Gadkari Memorial Diagnostic Centre offers high-quality MRI, CT scan,
In what can only be described as a transformative turning point for India’s medical technology sector, India Health 2025, hosted by Informa Markets at Pragati Maidan, New Delhi, has emerged as more than just a healthcare exhibition — it is a testament to India's medtech
The second edition of the India Health Exhibition 2025, organised by Informa Markets in India, was inaugurated today at Bharat Mandapam, New Delhi, highlighting the rising demand and immense potential in this sector. The three-day event, which concludes on 13th July 2025, returns with renewed
Andhra Pradesh is now home to its first-ever CDSCO‑notified auditing body for medical devices. In a significant move, the Central Drugs Standard Control Organisation (CDSCO) under Minister of Health and Family Welfare, Govt. of India has notified KIHT Certification Services (KCS) as the state’s first
Policy Mahotsav 2.0, held on 23rd May 2025 at the Constitution Club of India, concluded with resounding success, reinforcing India’s pivotal role in shaping the global MedTech narrative. Centered on the theme “Policy, Regulation, and Public Good: Building the MedTech Industry for Tomorrow’s Healthcare,” the
A national conference focusing on "Enhancing Pharmaceutical Quality Assurance through Good Manufacturing Practices (GMP)" brought together policymakers, industry leaders, and regulators at Andhra Pradesh MedTech Zone (AMTZ) today. Organized by the PHD Chamber of Commerce and Industry (PHDCCI) in collaboration with the Department of Pharmaceuticals,
In a ground-breaking achievement for India’s medical diagnostics sector, Erba Transasia proudly announces the commercial launch of the country’s first indigenously manufactured MonkeyPox PCR test kit. Validated by the Indian Council for Medical Research (ICMR), this kit today received emergency approval from Central Drugs Standard
Fischer Medical Ventures Limited (Fischer MV) is leading a paradigm shift in the healthcare sector by pioneering cutting-edge, cost-effective medical imaging technologies and state-of- the-art diagnostic innovations on a global scale.Headquartered in Chennai, India, with a manufacturing facility in the Andhra Pradesh MedTech Zone (AMTZ)
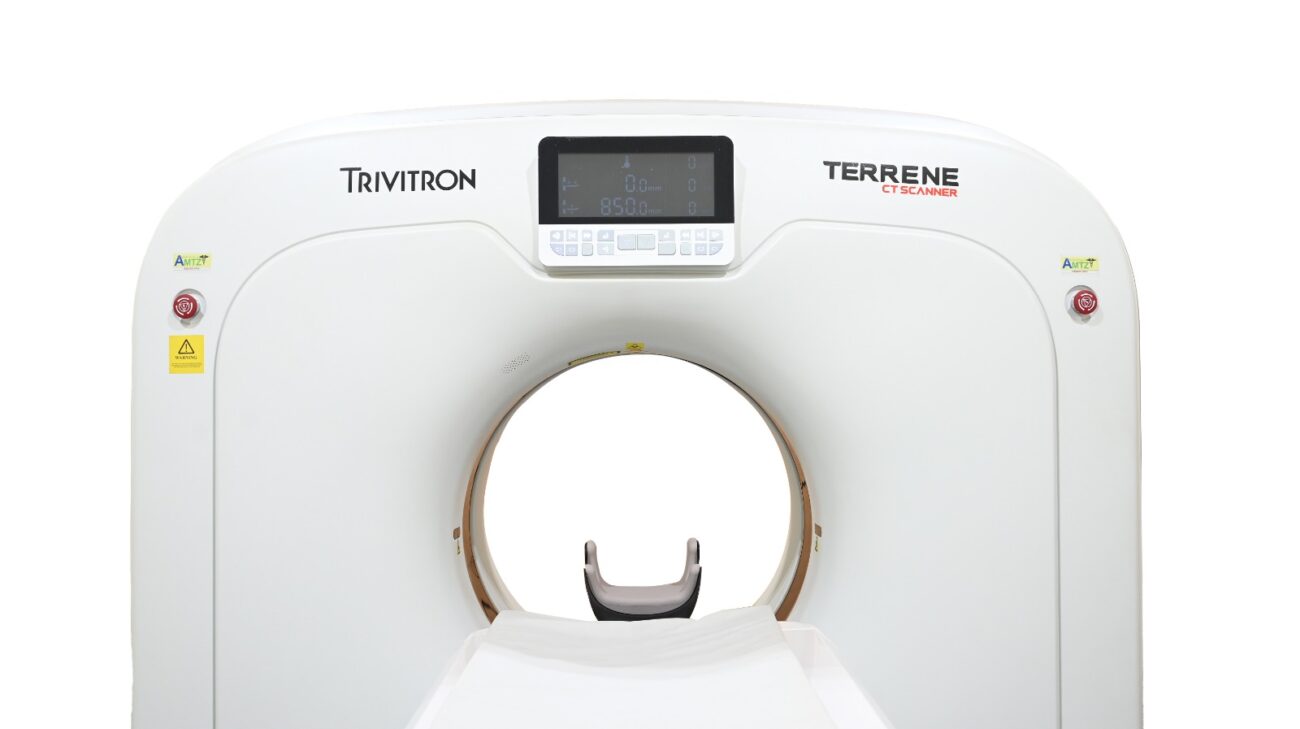
Trivitron Healthcare, a leading global medical technology company, proudly announces the launch of its state-of-the-art CT scanner, branded as “Terrene.” This ground-breaking technology is the first of its kind in India to receive approvals fromthe Bureau of Indian Standards (BIS), the Atomic Energy Regulatory Board