Medicover Hospital Performs Navi Mumbai’s First-Ever MyClip Procedure, Saves High-Risk Heart Patient Without Open SurgeryDeemed extremely high risk for repeat open-heart surgery due to weak heart function, the patient was treated using a heart-team approach with angioplasty followed by a minimally invasive MyClip procedure, offering
The Centre has worked with over 20 startups and 35 industry partners; providing end-to-end support ranging from industrial design and rapid prototyping to machining, post-processing and production.Strengthening India’s journey towards self-reliance in medical device manufacturing, the Advanced Additive Manufacturing Centre of Excellence (Coe) at Andhra
New Delhi, December 18, 2025: J Mitra & Company, one of India’s leading in-vitro diagnostics (IVD) manufacturers, has been conferred the Platinum Award in the Product Category – Medium Scale at the CII MedTech Quality Excellence Awards 2025, powered by the Andhra Pradesh MedTech Zone
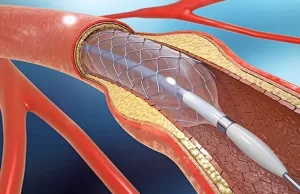
In a remarkable milestone for India’s medical device industry, a next-generation heart stent developed in India has received major global recognition, highlighting the country’s growing innovation and self-reliance in advanced cardiovascular technologies.The indigenous stent, designed and manufactured under the ‘Make in India’ initiative, has been
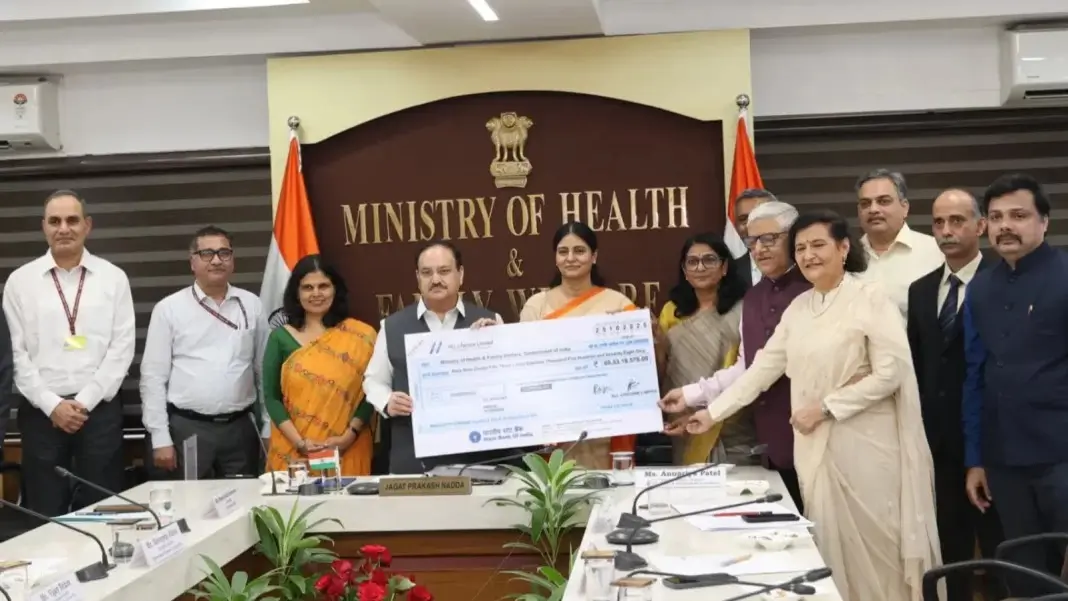
HLL Lifecare Limited (HLL), a Miniratna public sector enterprise under the Ministry of Health and Family Welfare (MoHFW), has paid a dividend of ₹69.53 crore to the Government of India for the financial year 2024–25, reaffirming its strong financial performance and contribution to India’s public
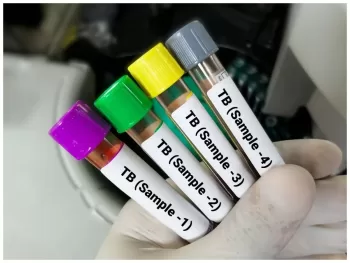
ICMR validates indigenous innovations that drastically cut testing time and cost, accelerating India’s goal to eliminate tuberculosis by 2025In a major leap toward India’s mission to eliminate tuberculosis (TB), the Indian Council of Medical Research (ICMR) has validated two new Made-in-India diagnostic tests developed by
The Government of India has announced the launch of the PRIP (Promoting Research and Innovation in Pharma-MedTech) scheme, inviting proposals from industry players, startups, and research organizations for high-impact projects aimed at strengthening India’s pharmaceutical and medical technology ecosystem. The initiative is designed to accelerate
India’s healthcare stakeholders — including hospitals, diagnostic chains, medical device firms, insurers and pharma groups — have responded positively to the rollout of GST 2.0, saying the tax changes announced under Prime Minister Narendra Modi’s leadership will alleviate cost pressures, expand access, and give a
Visakhapatnam, September 15, 2025: In a landmark moment for India’s MedTech ecosystem, Dr. Ajay Sood, Principal Scientific Advisor to the Hon’ble Prime Minister of India Shri Narendra Modi, officially released the M.Tech and MBA curriculum of Erba Global MedTech University, Visakhapatnam.The ceremony was graced by
“For a Viksit Bharat by 2047, India cannot limit itself to modest goals: The MedTech sector is a natural candidate for Atmanirbharata,” said Shri Piyush Goyal, Hon’ble Minister of Commerce and Industry at the 17th CII Global MedTech Summit 2025“The theme ‘Innovating for a Healthier