Researchers also concluded that a long gap after the second dose correlated with a higher chance of infection during the third wave Seventy per cent of the people who received a booster dose of the Covid vaccine did not contract the disease during the third wave,
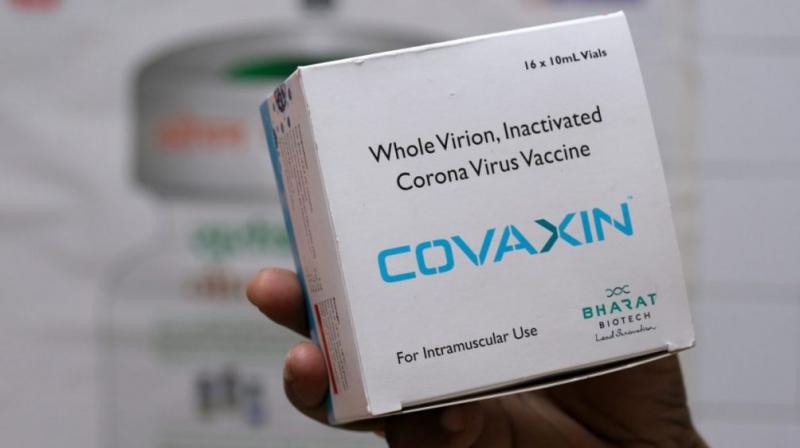
Bharat Biotech's Covaxin has been included to the approved list of COVID-19 vaccines for travel to Oman without Quarantine. In a tweet, Bharat Biotech noted: "Covaxin has now been added to the approved list of #COVID 19 vaccines for travel to Oman without quarantine. This will
According to the studies, A big drop in COVID antibodies was found within four months of vaccination
In a study of 614 fully vaccinated health workers in India discovered a "significant" drop in their COVID-fighting antibodies within four months of the first shot. This findings might help the Indian government to decide whether to provide booster doses. Some of the Western countries have done. The director
Union health minister Mansukh Mandaviya on Thursday said the government has launched an inquiry into a report claiming that counterfeit versions of Covishield, India’s primary anti-COVID-19 vaccine, had been seized. “It has been claimed that duplicate versions of Covishield vaccine were sold in the country. The
The Indian government has placed the largest order of vaccines so far. According to reports, it is procuring 660 million doses of Covaxin and Covishield the estimated worth of which is 145.05 billion Indian Rupees. This would help it meet the projections it has made
Individual member states of the European Union will have the option to accept vaccines authorised by the World Health Organisation (WHO) like Covishield for obtaining the bloc’s digital COVID certificate or ‘Green Pass’, an EU official said on Tuesday. The official said the European Medicines Agency
Fulfilling its commitment, Serum Institute of India has produced over 10 crore doses of its COVID-19 vaccine Covishield in June so far as India ramps up the pace of its vaccination drive in the face of a possible third wave of the viral infection. India’s COVID-19
A single dose of COVID-19 vaccine is sufficient for an infected person as there was a greater antibody response when compared with those who have had no prior infection, according to a study released by AIG Hospitals on Monday. The city-based AIG Hospitals recently published a
The government Friday said there is no need to panic on the need for an immediate change in the dosage interval of Covishield vaccine, underlining that reducing the time gap requires proper scientific study in the Indian scenario. In the context of some media reports quoting
Domestic vaccine major Serum Institute of India is seeking indemnity from liability for its COVID vaccines, saying that the rules should be same for all the companies, according to sources. The development comes after requests for indemnity and exemption from bridging trials have been made
- 1
- 2