Trivitron Healthcare announces the development of Real-Time PCR based kit for Monkeypox Virus
Medical devices company Trivitron Healthcare announced that it has developed the Real-Time PCR-based kit for the detection of Monkeypox (Orthopoxvirus) virus. The World Health Organization's Country Office for India recently requested the help of the
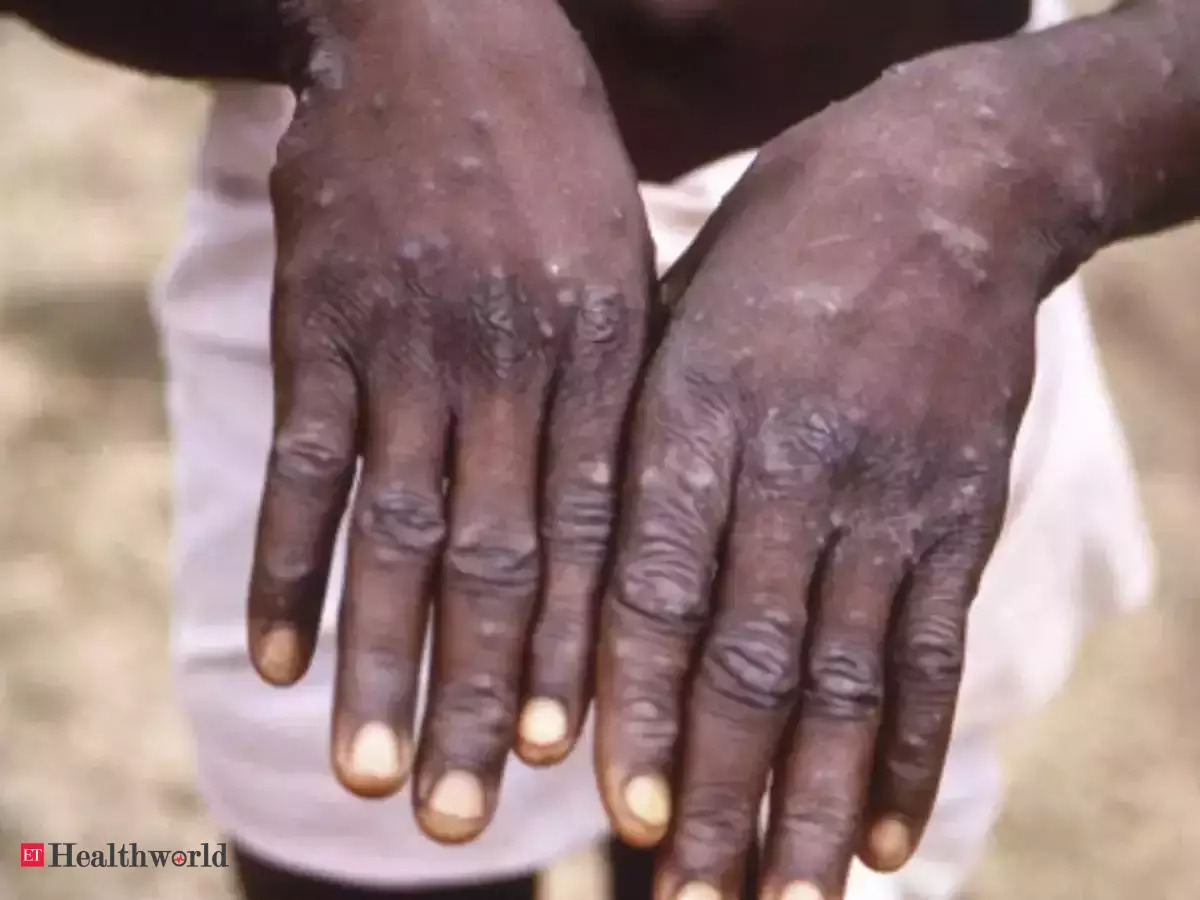
Medical devices company Trivitron Healthcare announced that it has developed the Real-Time PCR-based kit for the detection of Monkeypox (Orthopoxvirus) virus. The World Health Organization’s Country Office for India recently requested the help of the ICMR-National Institute of Virology in Pune to test suspected cases of Monkeypox for SEAR member-states. Monkeypox has become a disease of global public health importance, as it not only affects countries in West and Central Africa, but is also spreading at a fast pace globally.
For the response measures such as tracing efforts and treatment strategies, the diagnostic tools are crucial for responding to control the emerging public health challenges. Research & Development team of Trivitron Healthcare has developed a RT-PCR based kit for the detection of Monkeypox virus. Trivitron’s Monkeypox Real-Time PCR Kit is four colour fluorescence based kit, which can differentiate between Smallpox and Monkeypox in a one tube single reaction format, with total turnaround time of 1 hour. In this four gene RT-PCR kit, first target detects the viruses in the wider orthopox group, the second and third targets detects and differentiates the Monkeypox & Smallpox virus respectively and the fourth target detects the internal control corresponding to human cell to address the assay performance and aid in following its epidemic spread. This kit is available as Research-Use only (RUO) and based on literature search and in-silico design.
The World Health Organization (WHO) shares a ‘Nucleic Acid Amplification Testing’ is the preferred research laboratory technique for detection based on its sensitivity and specificity. As Orthopoxviruses are ‘serologically cross-reactive’, and antigen (Ag) and antibody (Ab) detection approaches do not provide Monkeypox – specific detection. Therefore, serology and antigen detection methods are not endorsed for detection or case investigation where resources are limited.
The WHO recommended specimen type for laboratory confirmation of Monkeypox is skin lesion material, including swabs of lesion surface and/or exudate, roofs from more than one lesion, or lesion crusts. Hence, both dry swabs and swabs placed in VTM can be used.
In the year 2017, Nigeria has experienced a large epidemic, with over 500-suspected cases of Monkeypox, and over 200 confirmed cases and a case fatalness ratio of about 3%. The cases continue to be reported until today. In May 2022, several suspected cases of Monkeypox were identified in quite a few non-endemic nations. Researches are currently underway to further understand the epidemiology, sources of spread, and the patterns of transmission of the virus.
Chandra Ganjoo, Group Chief Executive Officer, Trivitron Healthcare, informed that “Trivitron is committed to contribute cutting edge technologies for better screening of various ailments/ diseases. As a responsible MedTech product manufacturer, we feel that the current situation calls for immediate measures to stop the viral spread. India has always been at the forefront of extending help to the world, especially during the COVID-19 pandemic, and this time also the world needs assistance.”