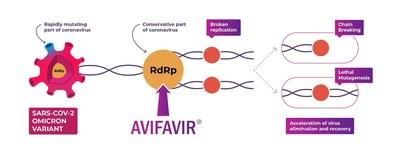
The conference saw participation from more than 1900 individuals, with over 285 eminent speakers sharing their experiences The Indian Society for Clinical Research (ISCR), an association of clinical research professionals, successfully hosted its 19th Annual Conference bringing together more than 1900 individuals and 285 eminent speakers to explore how digital
POST TAGS:
AIIMSBatra HospitalClinicalDr. Dangs LabDr. Ganesh DakhaleDr. Jerin JoseDr. Seema PaiDr. Sudeep GuptaDr. Suman KaranthDr. Upendra KaulDr. Y K GuptaFortisFortis Memorial Research CenterhealthcarehighlightedIndian Council of Medical ResearchPfizerpharmaceuticalSun PharmaTata Memorial Centretechnologies