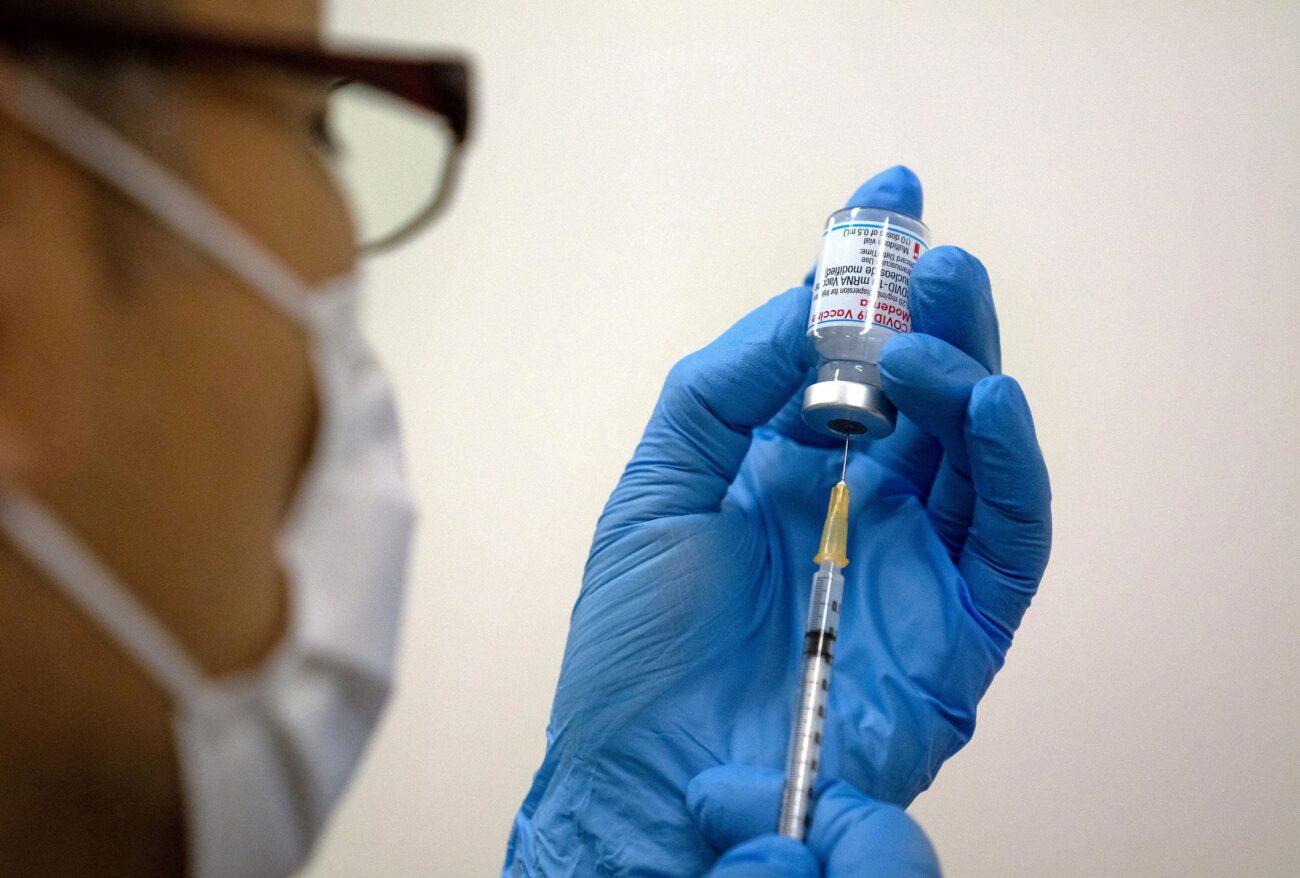
On Monday, Moderna Inc (MRNA.O) said its COVID-19 vaccine which generated a strong immune response in children aged six to 11 years and that it plans to submit the data to global regulators soon. Moderna said its two-dose vaccine generated virus-neutralizing antibodies in children and
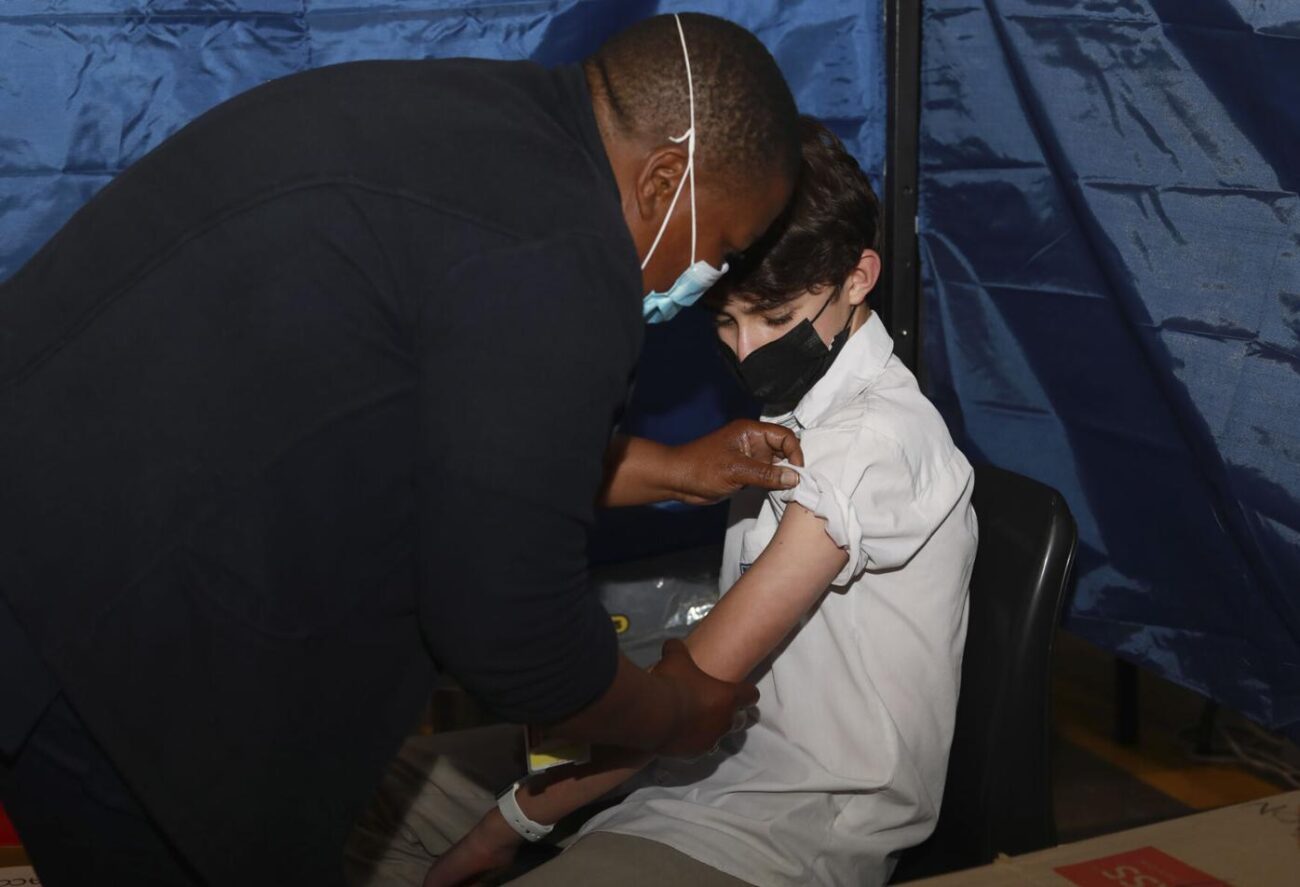
Vaccines for kids between the ages of 5 and 11 will likely be available in the first half of November, top U.S. infectious disease expert Anthony Fauci said on Sunday, predicting a timetable that could see many kids getting fully vaccinated before the end of
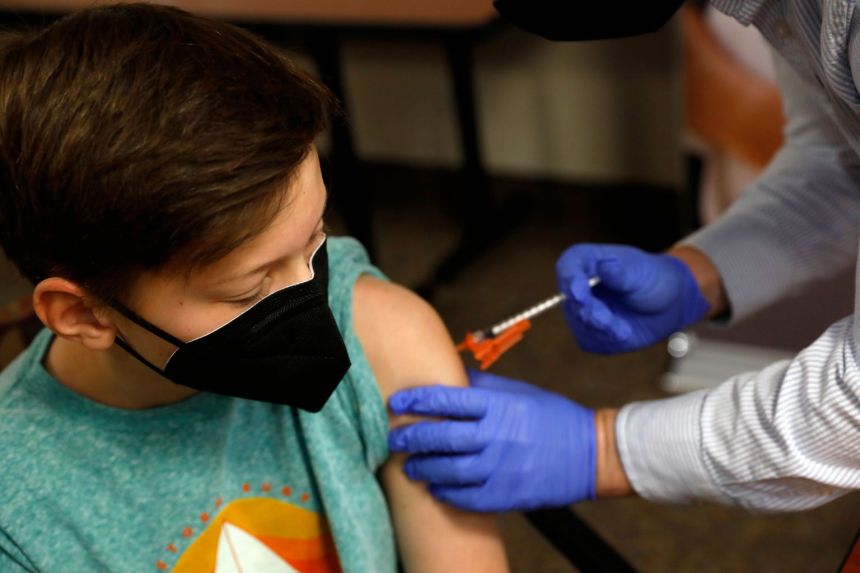
According to study details released Friday as the U.S. considers opening vaccinations to that age group, kid-size doses of Pfizer's COVID-19 vaccine appear safe and nearly 91per cent effective at preventing symptomatic infections in 5- to 11-year-olds. The shots could begin in early November with
South Africa has started giving COVID-19 vaccinations to adolescents aged between 12 and 17 years, with a goal of inoculating at least 6 million people from this age group. The country is trying to increase its rate of vaccinations which is running behind the target of
Various organizations in the U.S. are working to invest $3 billion in the vaccine supply chain as the country continues to position itself as a leading supplier of vaccines for the world, a top U.S. health official said on Thursday. The funding, which will begin to
First and only minimally invasive, transcatheter treatment specifically approved for premature babies with patent ductus, a life-threatening opening in their heart (a common congenital defect) One-fifth of 240,000 children born in India with congenital heart defects need early intervention to survive the first year