Punjab Health Minister Dr. Balbir Singh has announced the expansion of coverage under the state's healthcare scheme, a move aimed at improving access to specialized medical treatment and reducing the burden on patients who often travel long distances to major government hospitals for care. The decision
Vaping is increasingly gaining popularity among teenagers and young adults worldwide, but healthcare experts are warning that the trend may carry serious health risks despite being widely perceived as a safer alternative to traditional cigarettes. Electronic cigarettes, commonly known as e-cigarettes or vapes, are battery-operated devices
Unsafe food continues to pose a significant global public health threat, causing an estimated 1.5 million deaths every year worldwide, according to a new assessment released by the WHO. The findings are based on an analysis of 194 countries between 2000 and 2021, highlighting the persistent
Protein has long been considered the cornerstone of muscle building, but questions remain about how much is actually needed to maximize muscle growth. While strength training provides the stimulus for muscle development, nutrition experts say adequate protein intake is equally important for muscle repair, recovery,
Intermittent fasting has emerged as one of the most popular dietary trends worldwide, praised for its potential to support weight loss, improve metabolic health, and reduce the risk of chronic diseases. But as its popularity continues to grow, health experts say the science behind intermittent
For years, many people have believed that eating several small meals throughout the day can boost metabolism, promote weight loss, and improve overall health. However, emerging research suggests the answer may be more complex, with experts increasingly emphasizing meal quality and total calorie intake over
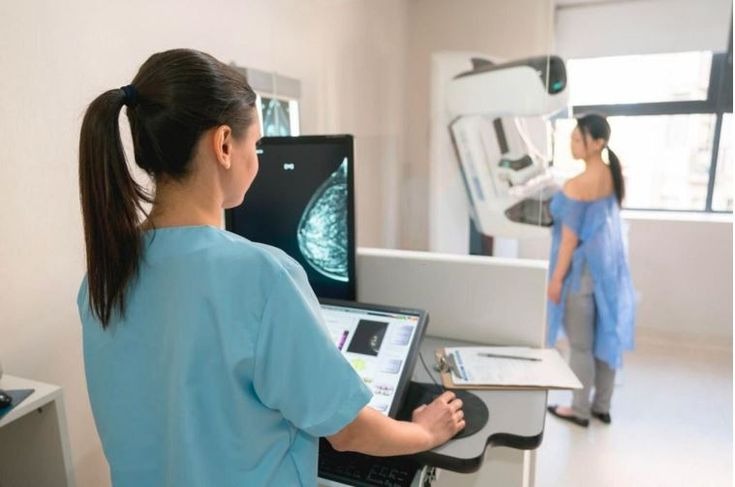
Updated breast cancer screening recommendations released by the American College of Physicians (ACP) have reignited discussions among healthcare professionals about the ideal age and methods for breast cancer screening, leaving many women uncertain about when they should begin routine mammograms. The revised 2026 guidance includes updated
Community Health Systems (CHS) has completed the $110 million sale of four Arkansas hospitals to Freeman Health System, marking a significant expansion for the regional healthcare provider and strengthening healthcare access across the Arkansas Missouri region. The acquisition effectively doubles Freeman Health System's hospital footprint and
As temperatures continue to soar across India, health conscious consumers are increasingly turning to chia seed water as a natural way to stay hydrated. Often described as one of the season's most popular wellness drinks, chia seed water is gaining attention not only for its
A baby's laughter may be more than just a heartwarming moment for parents it could offer valuable insights into early brain development, social learning, and emotional growth, according to emerging research in developmental neuroscience. Scientists studying infant behaviour have found that laughter plays a crucial role